OCRA
OCRA
Flow Diagram
The OCRA process Figure 6 has successfully been applied to treated PFAS-impacted waters for a wide range of long and short chain PFAS, in addition to co-contaminants such as metals, pesticides, hydrocarbons, and heavy natural organic matter or waste.

OCRA often achieves >99.9% removal of PFOS and PFOA per chamber, in a multi-chamber configuration. With the addition of the foam concentration system OSCAR (Ozofractionation Super Concentrating Array), the PFAS concentrate can represent <0.2% - 0.02% of the initial volume treated. The OSCAR super-concentrate can then be destroyed via a destructive process such as incineration, sonolysis, or electrochemical oxidation.
OCRA can act as a primary treatment stage to protect polishing technologies and when used with a polish, OCRA can achieve low ng/L or no detect levels of total PFAS in treated water.
The OCRA process utilises primary and secondary acidic ozofractionation chambers, often followed by an alkaline chamber. The OCRA system will treat co-contaminants such as petroleum hydrocarbons and chlorinated solvents by chemical oxidation.
The OCRA technology utilises multiple simultaneous treatment mechanisms for mixtures of contaminants: (1) foam fractionation for PFAS and other surface active compounds (2) an advanced oxidation process (AOP) using ozone for organic contaminants (3) a precipitation process resulting from redox potential ORP/pH adjustment for metals, followed by foam flotation and/or filtering (4) sedimentation / flotation for particulates. Each of these processes is reviewed in detail below.
Foam
Fractionation
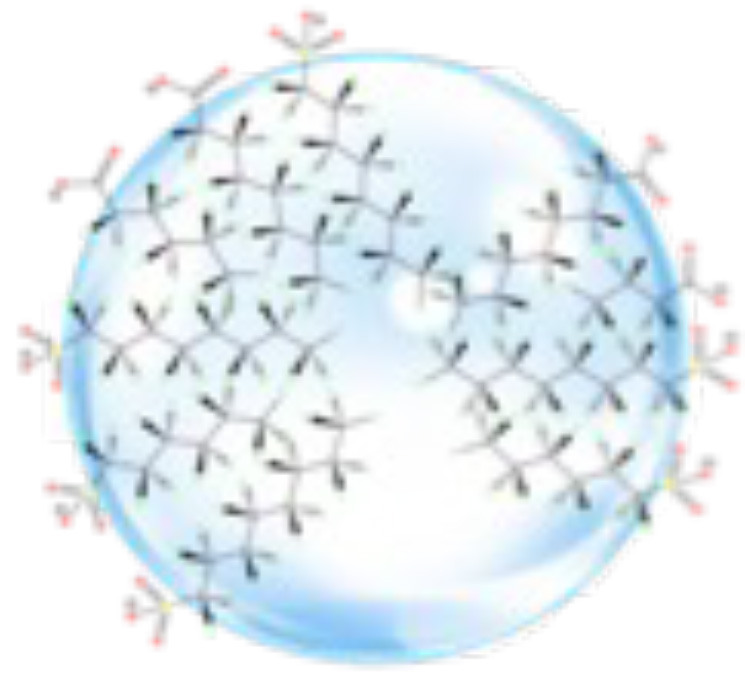
The OCRA process includes a series of chambers in which ozone is sparged from the base of the vessels through PFAS-impacted water to facilitate gas: water interface partitioning of dissolved PFAS into a foam fraction, which is collected from the surface of vessels as a PFAS concentrate (Dickson 2013, 2014) (Figure 7).
Efficient removal of PFAS via partitioning into gas bubbles has been demonstrated in the peer-reviewed literature (Lee et al. 2017; Meng et al. 2018), and the affinity of PFAS for the air: water interface is likewise central to their functionality in AFFF. The separation of PFOS and PFOA into foam with the assistance of ionic species has been shown to be an effective method for the recovery of fluorinated surfactants from dilute water solutions, with pH adjustment applied for optimisation resulting in demonstrated removal efficiencies of >99% (Lee et al. 2017). Thus, the propensity of PFAS to migrate to the gas: water interface is exploited to enable effective PFAS treatment.
The strong hydrophobic interactions and low van der Waals interactions displayed by perfluoroalkyl chains dramatically increase the tendency of fluorinated surfactants to self-assemble in water and to collect at interfaces (Krafft et al. 1998; Riess, 2002). Perfluoroalkyl chains are significantly more hydrophobic than their hydrogenated analogs and tend to attract via a fluorophilic mechanism, forming a fluorous phase. As PFAS populate the bubble interface, they align and self-assemble to form a monolayer (Gladysz et al. 2004), with the perfluoroalkyl tails lining up adjacent to each other inside the bubble and the anionic heads on the exterior of the bubble (Figure 7).



Ozone is the ideal gas for removal of PFAS from water, relative to other gases such as air, because of its propensity to form stable MNB emulsions, which have a very high surface area available for PFAS partitioning. Ozone bubbles have increased electrostatic repulsion between adjacent bubbles, or zeta potential, relative to oxygen, air, or nitrogen (Hu et al. 2018). The higher the zeta potential, the greater the stability of a bubble emulsion (Meegoda et al. 2018). Ozone MNB zeta potential can also be manipulated as it is dependent on solution pH (Hasegawa et al. 2009). Managing the pH and resulting zeta potential enables stable, long-lived ozone MNBs to form, maximising the interface surface area for very effective removal of PFAS from solution using ozofractionation.
Enrichment factors for PFOS and PFOA at the air: water interface were reported as 158 and 63, respectively (Psillakis 2009), with similar values reported elsewhere (Downes et al. 1995; Vecitis et al. 2008; Lunkenheimer et al. 2015). The limiting factor for removal of PFAS is the available gas: liquid interface surface area. This gas: water enrichment effect can be further enhanced by adding reagents which increase the solution ionic strength to ‘salt out’ PFAS to bubble interfaces, with relative efficiency approximating the Hofmeister series (i.e., acetate is more effective at salting out PFAS than chloride, which is in turn more effective than nitrate) (Lee et al. 2016; Cheng et al. 2010). The inverse relationship between increasing ionic strength and decreasing critical micelle concentration (CMC) of surfactants has been observed for decades (Corrin et al. 1947; Schick, 1964; Emerson et al. 1965; Kresheck, 1975; Chattopadhyay, 1996).
The formation of hemi-micelles on surfaces can occur at surfactant concentrations at 0.1 to 1% of the CMC (Schwarzenbach et al. 2003), with ionic strength also influencing hemi-micelle formation. The ionic strength of seawater has a dramatic effect on the solubility of PFOS, which is reported to diminish from 750-680 mg/L to 12-25 mg/L from freshwater to seawater (Giesy et al., 2006; OECD, 2002). The change in partitioning properties of long chain PFAAs was demonstrated in an estuary where a halocline caused sediment water partitioning values to increase exponentially as a function of salinity. Due to this salting-out effect, PFAAs were largely scavenged by adsorption onto suspended solids and/or sediments in estuarine environments (Hong et al. 2013). In the same manner increases in ionic strength in the OCRA process causes a dramatically enhanced partitioning of PFAS to ozone bubble surfaces.
The combination of the reagent addition with its surfactant “salting out” effect and the large interfacial surface area of the MNB emulsion, creates an aqueous environment which propels PFAS to migrate and self-assemble at the interface of the ozone bubbles, which subsequently rise and are removed as a foam fraction. These combined effects result in a very effective method to collect and concentrate PFAS (Ross et al., 2017).
Advanced
Oxidation
Processes
Ozone is an effective oxidant for breaking down co-contaminants such
as sewage, natural organic matter, petroleum hydrocarbons and
chlorinated solvents, and it can result in partial conversion of
perfluoroalkyl acid (PFAA) precursors to PFAAs (Table S1). Ozone is a
powerful oxidant, forming hydroperoxide, hydroxyl and ozone radicals.
The ozone radical is preferentially generated at higher pH (Suthersan et
al. 2017).
Treatment of non-PFAS organic co-contaminants is a distinct advantage of OCRA, allowing oxidative treatment and mineralisation of co-contaminants such as hydrocarbons and chlorinated solvents, which are usually subject to discharge criteria. Oxidation of these organic co-contaminants also reduces the competitive load of constituents that can compete with PFAS for adsorption sites if an adsorption polish is used to achieve very low discharge limits. The use of ozone also manages sulfides, which may also be present in anaerobic groundwater in fire training areas, as sulfides are oxidised to the more innocuous sulfate. The inlet flow rates and percentage ozone added to individual ozofractionation chambers can be adjusted to allow chemical oxidation of co-contaminants to proceed, as required depending on the co-contaminant loading.
Metal
Precipitation
The removal of dissolved, reduced metals by oxidation and pH precipitation is achieved using OCRA as a result of the oxidative conditions of the fractionation chambers in combination with pH adjustment. Metals are precipitated to form insoluble metal oxide species and are either removed by physical filtration or within the foam
fractions. Ozofractionation has been demonstrated to remove aluminium, arsenic, cadmium, lead, copper, cobalt, iron, manganese, nickel and zinc.
These precipitated metals can also have a beneficial effect as a removal mechanism for dissolved PFAS, which adhere to their surfaces (Park et al, 2018). Dissolved iron and manganese are commonly found in anaerobic groundwater associated with hydrocarbon impacts. Because they tend to precipitate out when pumped to the surface, they can foul GAC and IX resins. Ozofractionation, which removes iron and manganese as oxide precipitates, allows management of these interfering species. The OCRA process also removes solid particles based on their density, with dense solids falling to the base of the vessel for removal and less dense material floated to the surface for removal with the foam fraction and filtering.
Ozofractionation
Super Concentrating
Array (OSCAR)
The OCRA system also contains a dedicated ozofractionation chamber for concentrating the foam fraction from the multiple 1st pass ozofractionation stages, followed by a vacuum assisted dehydration unit, which are together termed OSCAR. The vacuum assisted dehydration process uses waste heat from other system components and excess vacuum available in the OCRA process. OSCAR processes the 1st pass fractions from the OCRA system to reduce foam volumes by >95%. Using the OSCAR system as part of the OCRA process can potentially reduce the volume of waste foam concentrate to less than 0.2% of the original volume of the impacted water. The concentrate can then be destroyed via a destructive process such as incineration, sonolysis, or electrochemical oxidation.
